Sign Up for Updates
Stay informed and stay current with updates delivered directly to your inbox.
Congenital athymia is a very rare primary immunodeficiency in infants born without a functional thymus, rendering them highly immunodeficient.1
The precise incidence of congenital athymia is difficult to estimate, as it has often been reported as a clinical feature across many disease syndromes rather than an isolated diagnosis.2
While congenital athymia may be idiopathic, individuals often have associated genetic or teratogenic disorders, such as2-5:
cDGS, complete DiGeorge Syndrome; CHARGE: coloboma, heart defects, atresia of the choanae, retardation of growth and mental development, genital defects, and ear defects and hearing loss; FOXN1, forkhead box N1; US: United States
1. Hsieh EWY, Kim-Chang JJ, Kulke S, et al. Defining the Clinical, Emotional, Social, and Financial Burden of Congenital Athymia. Adv Ther. 2021;38(8):4271-4288. doi:10.1007/s13235-021-01820-9. 2. Collins C, Sharpe E, Silber A, et al. Congenital Athymia: Genetic Etiologies, Clinical Manifestations, Diagnosis, and Treatment. J Clin Immunol. 2021;41(5):881-895. doi:10.1007/s10875-021-01059-7. 3. Mansouri N, Khezripour E, Rashtiani N, et al. Gestational diabetes and its effect on fetal thymus size: a case-control study. BMC Pregnancy Childbirth. 2025;25(1):330. Published 2025 Mar 21. doi:10.1184/s12884-025-07468-5. 4. Lammer EJ, Chen DT, Hoar RM, et al. Retinoic acid embryopathy. N Engl J Med. 1985;313(14):837-841. doi:10.1056/NEJM198510033131401. 5. Biggs SE, Gilchrist B, May KR. Chromosome 22q11.2 Deletion (DiGeorge Syndrome): Immunologic Features, Diagnosis, and Management. Curr Allergy Asthma Rep. 2023;23(4):213-222. doi:10.1007/s11882-023-01071-4. 6. Cortés-Martín J, Peñuela NL, Sánchez-García JC, et al. Deletion Syndrome 22q11.2: A Systematic Review. Children (Basel). 2022;9(8):1168. Published 2022 Aug 3. doi:10.3390/children9081168. 7. Szczawińska-Popłonyk A, Schwartzmann E, Chmara Z, et al. Chromosome 22q11.2 Deletion Syndrome: A Comprehensive Review of Molecular Genetics in the Context of Multidisciplinary Clinical Approach. Int J Mol Sci. 2023;24(9):8317. Published 2023 May 5. doi:10.3390/ijms24098317. 8. Biggs SE, Gilchrist B, May KR. Chromosome 22q11.2 Deletion (DiGeorge Syndrome): Immunologic Features, Diagnosis, and Management. Curr Allergy Asthma Rep. 2023;23(4):213-222. doi:10.1007/s11882-023-01071-4.
The thymus plays an essential role in T-cell development. In a healthy immune system, bone marrow-derived T-cell precursors migrate to the thymus where they undergo positive and negative selection, resulting in immunocompetent, naïve T cells capable of mounting a balanced immune response.
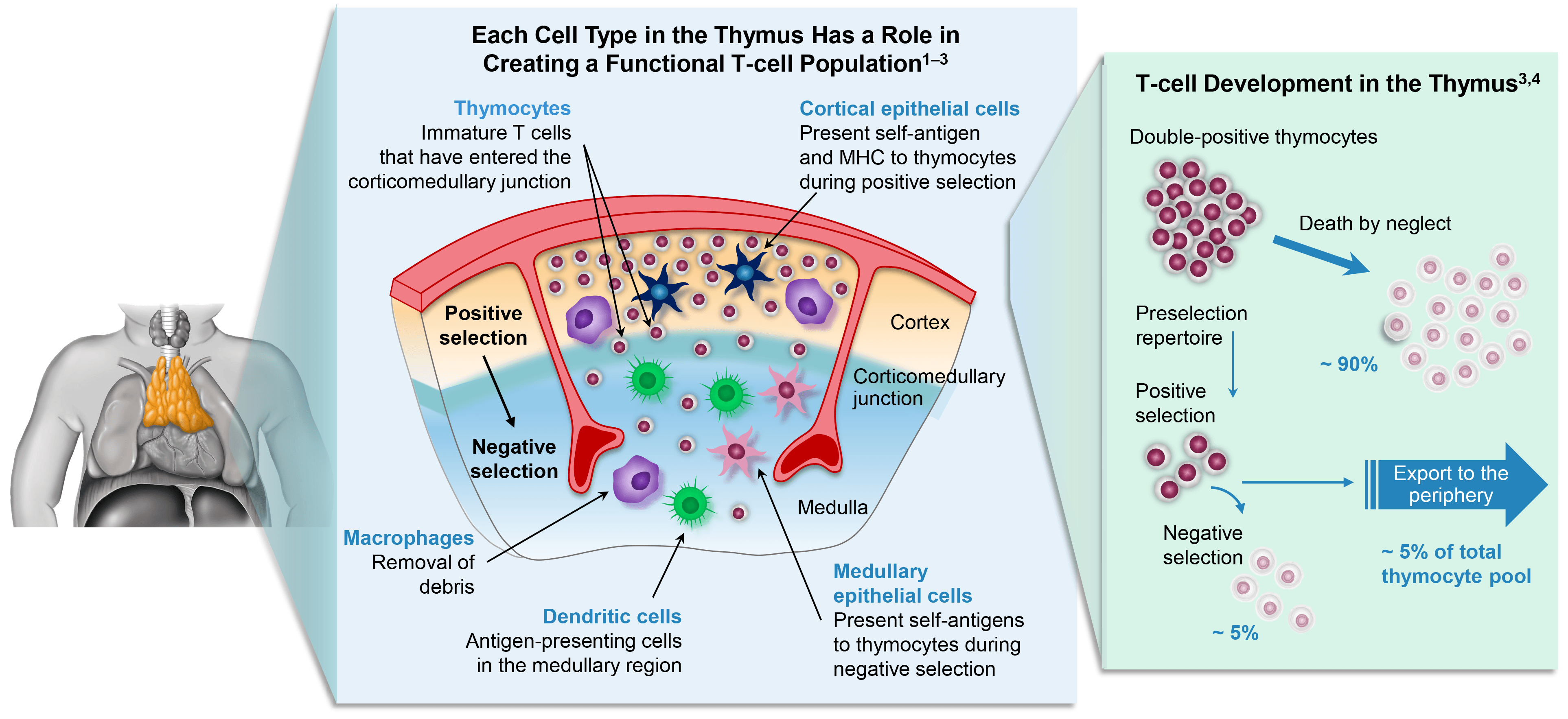
In patients with CA, the absence of functional thymic tissue leads to a disruption in the pathway, resulting in T-cell precursors being unable to mature into immunocompetent T cells.
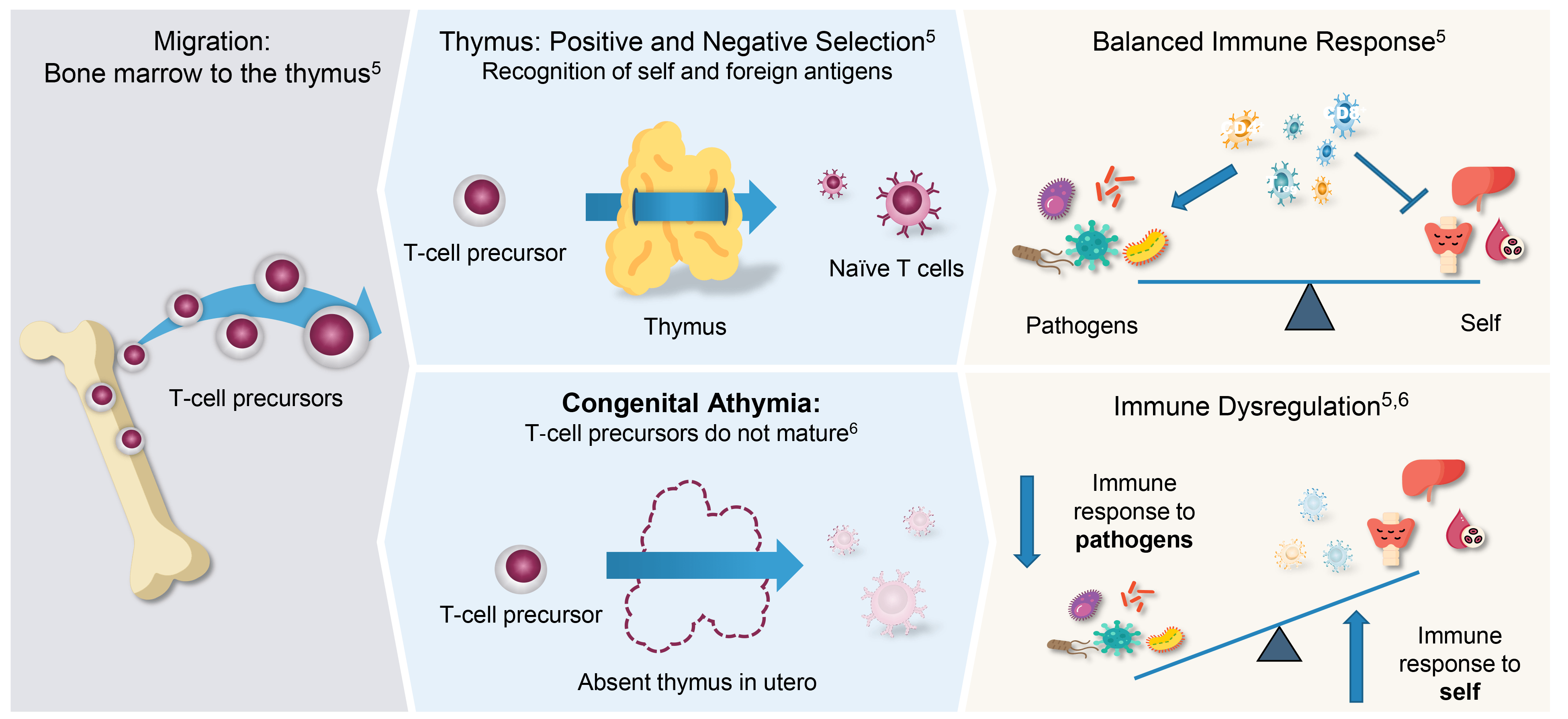
As a result of the lack of immunocompetent T cells, patients with CA present with clinical manifestations such as:
CA, congenital athymia; GVHD, graft-versus-host disease
1. Alexandropoulos K, Danzl NM. Immunol Res. 2012;54:177-190; 2. Klein L, et al. Nat Rev Immunol. 2009;9:833-844; 3. Love PE, Bhandoola A. Nat Rev Immunol. 2011;11:469-477; 4. Palmer E. Nat Rev Immunol. 2003;3:383-391; 5. Lewis DE, Blutt SE. In: Clinical Immunology: Principles and Practice. 5th ed. Elsevier; 2019:19-38.e1; 6. McGhee SA, et al. Immunol Res. 2009;45(1):37-45; 7. Collins C, Sharpe E, Silber A, et al. J Clin Immunol. 2021;41(5):881-895. doi:10.1007/s10875-021-01059-7.
Patients with CA are normally identified through newborn screening for SCID in the US
Diagnosis of CA requires confirmation of low naïve T cell numbers through flow cytometry. B cell and NK cell numbers are not typically affected, meaning patients present with a T−B+NK+ phenotype
SCID patients may also present with a T−B+NK+ phenotype, so genetic evaluation is used to identify underlying genetic mutations
If no known mutations related to SCID or CA are identified, further testing may be required. The artificial thymic organoid (ATO) assay assesses whether hematopoietic stem cells are able to mature into T cells, aiding the differentiation between SCID and CA, depending on the results.1,4
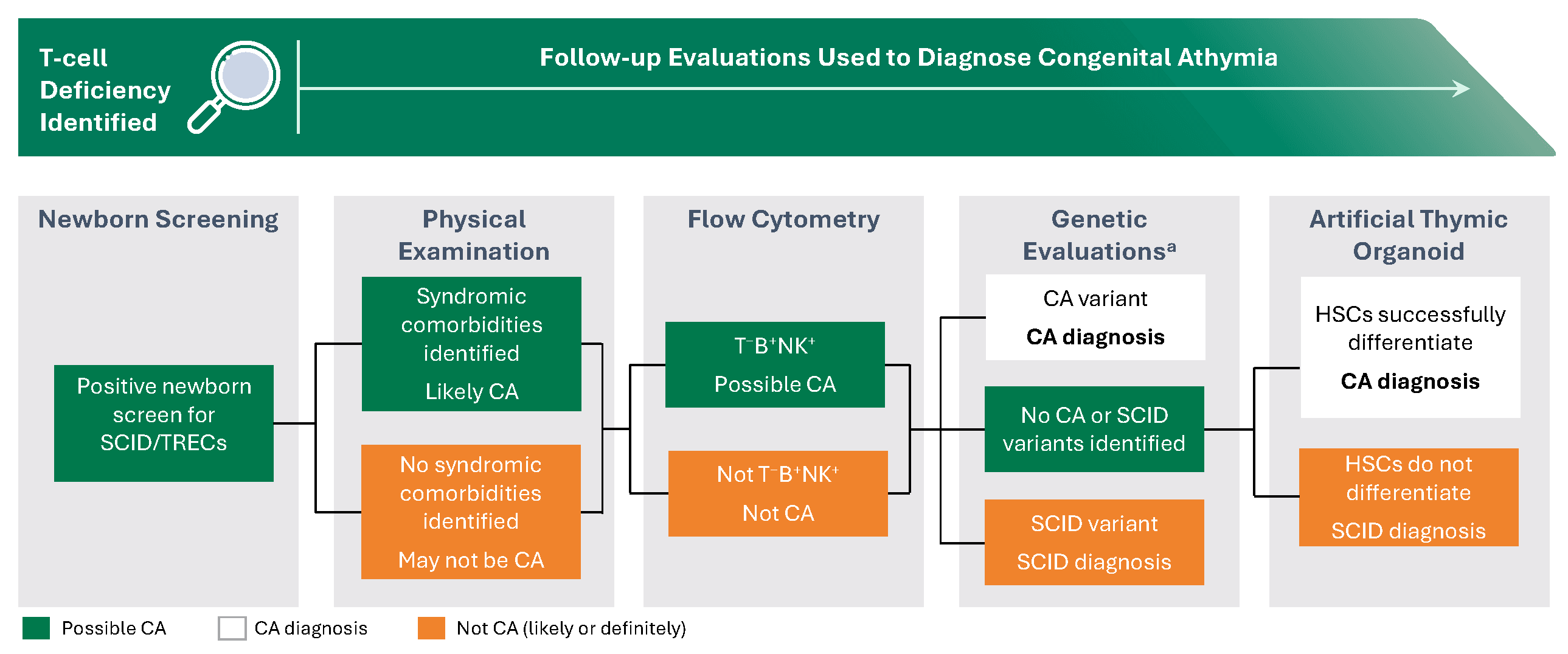
ATO, artificial thymic organoid; B, B-celll; CA, congenital athymia; HSC, hematopoietic stem cell; NK, nature killer; SCID, severe combined immunodeficiency; T, T-cell; TREC, T-cell receptor excision circle; US, United States
aInclude SCID genetic panels, chromosomal microarray, and whole genome/exome screening.
1. Collins C, Sharpe E, Silber A, et al. Congenital Athymia: Genetic Etiologies, Clinical Manifestations, Diagnosis, and Treatment. J Clin Immunol. 2021;41(5):881-895. doi:10.1007/s10875-021-01059-7. 2. Bifsha P, Leiding YW, Pai SY, et al. Diagnostic assay to assist clinical decisions for unclassified severe combined immune deficiency. Blood Adv. 2020;4(12):2606-2610. doi:10.1182/bloodadvances.2020001736. 3. Bosticardo M, Pala F, Calzoni E, et al. Artificial thymic organoids represent a reliable tool to study T-cell differentiation in patients with severe T-cell lymphopenia. Blood Adv. 2020;4(12):2611-2616. doi:10.1182/bloodadvances.2020001730. 4. Soulard C, Pala F, Bosticardo M, et al. Probing the diagnosis of SCID through in vitro T-cell development: Experience in 2 centers in North America. J Allergy Clin Immunol. 2025;156(4):906-908.
Stay informed and stay current with updates delivered directly to your inbox.
Submit a request for more information.
+ 1 (800) 739-0565Monday - Friday, 8 AM - 8 PM ET
Please report any side effects related to any Sumitomo Pharma America, Inc. medication by contacting us. You may also contact the US Food and Drug Administration (FDA) directly by visiting www.fda.gov/Safety/MedWatch or calling 1-800-FDA-1088.