Medical Science Liaisons
Find and connect with your Medical Affairs Contact, also known as Medical Science Liaison (MSL), in your region and specialty.
people are estimated to be affected in the US
of US adults aged 40 years and older report OAB symptoms
Women are more commonly affected by urgency, frequency, nocturia, and urgency urinary incontinence. Prevalence estimates are based on a population study of approximately 20,000 US adults. 1,3
OAB Symptom Frequency
Women
Men
Sometimes
43.1%
27.2%
Often
32.6%
15.8%
OAB, overactive bladder; US, United States.
1. Coyne KS, Sexton CC, Vats V, Thompson C, Kopp ZS, Milsom I. Urology. 2011;77(5):1081-1087. 2. US Census Bureau. Age and Sex Composition: 2023. Available at: https://www.census.gov/data/tables/2023/demo/age-and-sex/2023-age-sex-composition.html. Accessed April 28, 2026. 3. Mayo Clinic. Overactive bladder: symptoms and causes. Available at: https://www.mayoclinic.org/diseases-conditions/overactive-bladder/symptoms-causes/syc-20355715. Accessed April 28, 2026.
The process of micturition is a neural feedback process - as the bladder fills, stretch receptors send signals through a spinal pathway.
This normally will trigger the detrusor muscle of the bladder to contract and the internal urethral sphincter to relax once a certain volume is reached, resulting in an urge to urinate.
This reflex is primarily governed by parasympathetic autonomic activation, while urine storage and retention are more associated with sympathetic drivers.
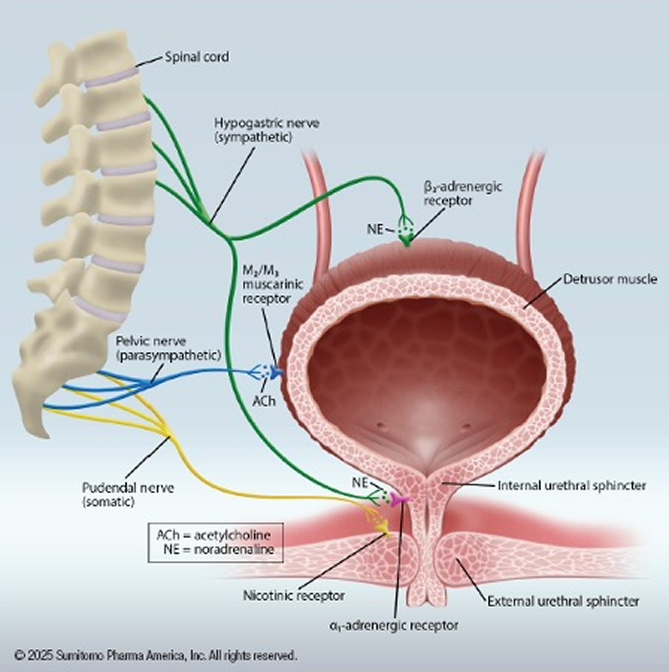
There are many causes of overactive bladder, but in general, OAB occurs when the detrusor muscle experiences frequent involuntary contractions associated with increased activation of the parasympathetic nervous system. This is modulated by heightened stimulation of muscarinic neuronal receptors and subsequent acetylcholine release from the parasympathetic nerve terminal. This causes relative overactivity of the detrusor muscle and the accompanying urinary urgency sensation.3
OAB, overactive bladder.
1. Fowler CJ, Griffiths D, de Groat WC. The neural control of micturition. Nat Rev Neurosci. 2008;9(6):453-466. 2. Verhamme KM, Sturkenboom MC, Stricker BH, Bosch R. Drug-induced urinary retention: incidence, management and prevention. Drug Saf. 2008;31(5):373-388. 3. Ellsworth PI, Kim ED. Overactive bladder [WebMD Medscape website]. January 27, 2020. Available at: https://emedicine.medscape.com/article/459340-overview#a5. Accessed April 28, 2026.
OAB is characterized by urinary urgency, usually accompanied by increased daytime frequency and/or nocturia, in the absence of urinary tract infection or other detectable disease. Patients may present with urinary incontinence (OAB-wet) or without incontinence (OAB-dry). While up to 7 micturition episodes during daytime may be considered normal frequency, the actual total can be highly variable based on sleep duration, fluid intake, comorbid medical conditions, and other factors.1,2
Nocturnal polyuria
Low nocturnal bladder capacity
Polydipsia
Diabetes insipidus
Urinary tract infection
Interstitial cystitis or bladder pain syndrome
Atrophic vaginitis (in females)
OAB, overactive bladder.
1. International Continence Society. ICS glossary of terminology. Available at: https://www.ics.org/glossary. Accessed April 28, 2026. 2. Cameron AP, Chung DE, Dielubanza EJ, et al. J Urol. Published online April 23, 2024. doi:10.1097/JU.0000000000003985. 3. Mayo Clinic. Overactive bladder: symptoms and causes [Mayo Clinic website]. Available at: https://www.mayoclinic.org/diseases-conditions/overactivebladder/symptoms-causes/ syc-20355715. Accessed August 18, 2020.
Find and connect with your Medical Affairs Contact, also known as Medical Science Liaison (MSL), in your region and specialty.
Stay informed and stay current with updates delivered directly to your inbox.
Submit a request for more information.
+ 1 (800) 739-0565Monday - Friday, 8 AM - 8 PM ET
Please report any side effects related to any Sumitomo Pharma America, Inc. medication by contacting us. You may also contact the US Food and Drug Administration (FDA) directly by visiting www.fda.gov/Safety/MedWatch or calling 1-800-FDA-1088.